 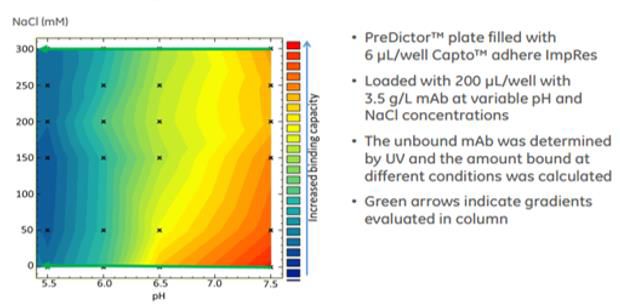
Multimodal chromatography uses small ligands with multiple modes of interaction, e.g., charged, hydrophobic or hydrogen bonding, to separate proteins from complex mixtures. Our approach presents a promising route to the engineering of multimodal patterns for future chromatographic resin design. Quantitative differences between the two suggest opportunities for improvements in the coarse-grained energy function for efficient predictions of cluster and pattern formations. Despite their simplicity, the coarse-grained simulations qualitatively capture the cluster size distribution of ligands observed in detailed MD simulations. To explore how these trends in ligand association extend to a larger surface with high density of ligands, we performed coarse-grained Monte Carlo (MC) simulations of a 132-ligand surface using ligand interactions parametrized based on free energies obtained from the three-ligand MD simulations. We find that although the formation of a contact between two neighboring ligands is slightly unfavorable, three neighboring ligands exhibit a preference for the formation of a fully connected cluster. Furthermore, we find that tethering a ligand to a surface restricts its conformations to a subset of those observed in free solution, yet the ligand maintains flexibility in the plane of the surface and can form contacts with neighboring ligands. Here, we study interfacial systems of increasing complexity-from a single immobilized multimodal ligand to high density surfaces-to better understand how ligand behavior is affected by the presence of a surface and the presence of other ligands in the vicinity, and how this behavior scales to more » larger systems. 
Recently, we used molecular dynamics (MD) simulations to show that ligands immobilized on surfaces can interact and associate with neighboring ligands to form hydrophobic and charge patches, which may have important implications for the nature of protein–surface interactions. Multimodal chromatography uses multiple modes of interaction such as charge, hydrophobic, or hydrogen bonding to separate proteins. This new understanding of multimodal surfaces has important implications for developing improved predictive models and designing new classes of multimodal separation materials. This clustering phenomenon is likely to play a key role in governing protein–surface interactions in multimodal chromatography. Finally, we developed an approach for quantifying differences in the observed surface patterns by calculating distributions of the patch more » size and patch length. Further, the use of flexible linkers enabled hydrophobic groups to collapse to the surface, reducing their accessibility. In addition, the introduction of a flexible linker (corresponding to the commercially available ligand) enhanced cluster formation and allowed aggregation to occur at lower surface densities. On the other hand, lowering the surface density to 1 ligand/3 nm 2 reduced or eliminated this aggregation behavior. For aggregating ligands, we report this resulted in the formation of a surface pattern that contained relatively large patches of hydrophobicity and charge whose sizes exceeded the length scale of the individual ligands. We found that ligands that were flexible and terminated in a hydrophobic group had a propensity to aggregate on the surface, while less flexible ligands containing a hydrophobic group closer to the surface did not aggregate. In this work, we performed molecular dynamics simulations of a series of multimodal cation-exchange ligands immobilized on a hydrophilic self-assembled monolayer surface at the commercially relevant surface density (1 ligand/nm2). Multimodal chromatography is a powerful tool which uses multiple modes of interaction, such as charge and hydrophobicity, to purify protein-based therapeutics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
